Our Quantum Advantage challenge with $20,000 BTC award is live.
More Details →
“If atoms could speak, their language would be poetry."
Niels Bohr's poetic observation marked the challenge of expressing the nuanced world of quantum mechanics.
Quantum mechanics, emerging in the 20th century, revolutionized our understanding of the microscopic.
Erwin Schrödinger's quantum mechanical model, a departure from fixed orbits, uses wave functions to describe electron probabilities. Join the journey from Bohr to Schrödinger, exploring a practical guide to electrons' behavior.
Visit BlueQubit to learn more about the quantum realm!
Quantum mechanics is a fascinating branch of physics that delves into the behavior of matter and energy at the smallest scales – think atoms and subatomic particles. It emerged in the early 20th century, revolutionizing our understanding of the physical world.

The wave functions of the electron in a hydrogen atom at different energy levels capture a core principle of quantum mechanics – probability.
According to Niels Bohr's observation, "When it comes to atoms, language can only be used as in poetry." This sentiment highlights the profound challenge of expressing the nuances of this microscopic world.
In the image below, the brighter areas indicate a higher probability of finding the electron. This doesn't mean the electron is definitively there; rather, it suggests the likelihood of locating it in those regions. This probabilistic nature is a fundamental departure from classical mechanics and challenges our intuition about the determinism of the physical world.
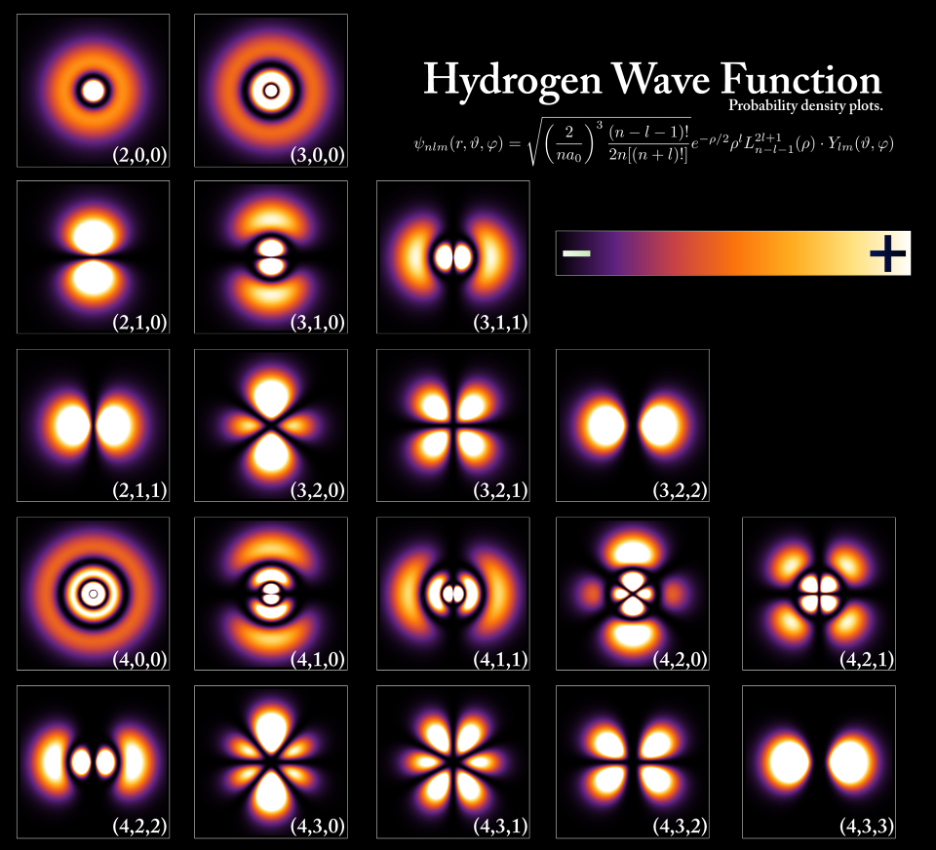
The quantum mechanical model describes particles not as tiny billiard balls with well-defined trajectories but as entities characterized by wave functions. The latters encapsulate the probabilities of a particle's properties, like its position and momentum. The famous Heisenberg Uncertainty Principle, a cornerstone of quantum mechanics, states that there's an inherent limit to the precision with which we can simultaneously know certain pairs of properties, such as position and momentum.
In the late 19th and early 20th centuries, the quest to unravel the mysteries of atomic structure led scientists through a series of evolving models. The journey started with the plum pudding model, only to face a setback with Ernest Rutherford's gold foil experiment, which contradicted the initial model. As electrons appeared destined to collide with the nucleus and cause annihilation, Rutherford's nuclear model raised questions about the stability of the atom.
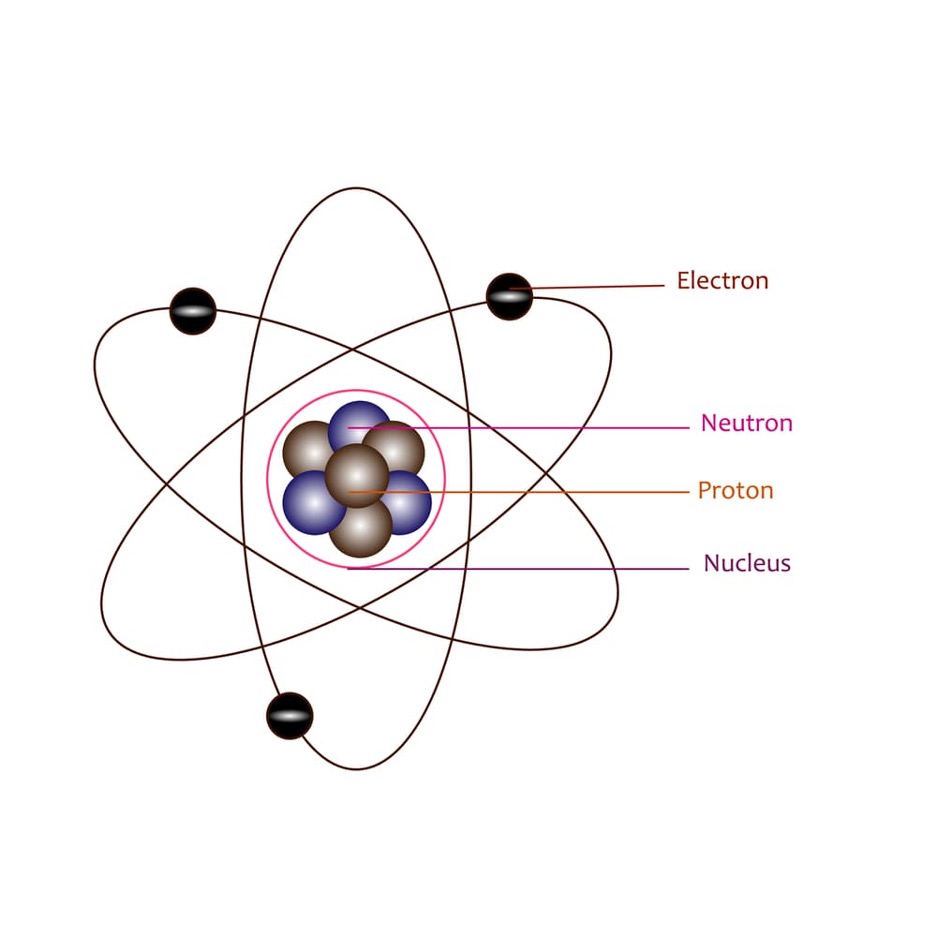
As the understanding of quantized energy emerged, thanks to Planck's quantum theory and Einstein's applications in Blackbody Radiation and the photoelectric effect, Niels Bohr introduced quantization to the angular momentum of electrons. Bohr's model successfully explained the hydrogen atom's spectra, but its limitations persisted with more extensive elements.
The wave-particle duality, encompassing both particle-like and wave-like behaviors of light, further deepened the understanding. Louis de Broglie's synthesis of Planck's and Einstein's equations revealed the wavelength nature of all matter.

The turning point arrived with Erwin Schrödinger's atomic theory work in 1926. He developed an intricate mathematical equation to describe the behavior of electrons in hydrogen atoms by taking advantage of the wave-particle duality. The quantum mechanical model of the atom was made possible as a result, and thus marked the beginning of quantum mechanics.
Unlike Bohr's model, the quantum mechanical model of the atom, derived from Schrödinger model, doesn't confine electrons to specific orbits. Instead, it introduces wave functions, representing the probability of finding electrons within an electron cloud. This cloud, visualized as the result of dropping a marker onto a paper, indicates varying densities, with higher probabilities closer to the nucleus.
"Orbital" refers to the 90% probability region within the electron cloud, embodying the evolution from classical quantum atom models to the profound world of quantum mechanics.
The quantum mechanical model of the atom revolutionized our understanding of atomic structure. So, let's explore its main features below:
Instead of fixed orbits, electrons are described by probability distributions called orbitals. These represent the regions where electrons are likely to be found.
Electrons can only exist in specific energy levels, and they transition between these levels by absorbing or emitting discrete amounts of energy in the form of photons.
Electrons exhibit both wave-like and particle-like behavior. The wavefunction captures this duality, a mathematical function that describes the probability amplitude of finding an electron at a particular position.
Formulated by Heisenberg, this principle states that it is impossible to know a particle's precise position and momentum simultaneously. The more accurately one is known, the less accurately the other can be determined.
These include the principal quantum number (n), azimuthal quantum number (l), magnetic quantum number (m_l), and spin quantum number (m_s). They describe various properties of electrons and their spatial and spin orientations.
Electrons have an intrinsic property called spin, with two possible values: up or down. The spin quantum number represents this and is crucial in understanding electron behavior.
No two electrons in an atom can have the same set of quantum numbers. This principle prevents electrons from occupying the same quantum state and is fundamental to the stability and structure of atoms.
The probability distribution of finding an electron is visualized as an electron cloud, representing the three-dimensional space where an electron is likely to be located.
These features collectively provide a comprehensive and accurate description of the behavior and properties of electrons in an atom, forming the foundation of quantum mechanics.
His atomic model is not just a theory; it's a practical guide to understanding the behavior of electrons in the quantum realm.
In this model, electron motion is unrestricted by orbital rigidity. The probability that electrons will be found within an electron cloud is represented by Schrödinger's wave functions. It provides a more flexible and accurate representation of electron activity than the rigid paths of classical models.
Examine the idea of orbitals, which are areas where electrons have a 90% chance of being present. In contrast to traditional models, which specify exact pathways, orbitals introduce some uncertainty. They give a useful foundation for comprehending where electrons might end up in an atom.
Schrödinger's equation introduces a quantized energy scale, a departure from continuous energy levels in classical physics. This practical aspect allows us to understand the distinct, quantized energy states of electrons in an atom.
Additionally, Schrödinger's name is often associated with the intriguing thought experiment known as "Schrödinger's cat." This experiment delves into the law of superposition, where a cat in a sealed box can exist simultaneously, alive and dead, until observed.
This concept adds philosophical depth to Schrödinger's contributions to quantum mechanics, exploring the profound implications of superposition and observation in the quantum realm. To visually explore Schrödinger's cat, check out this video:
Understanding the distribution of electrons within an atom is essential when learning quantum mechanics and electron configuration.
The Bohr model, though one-dimensional, utilized the quantum number 'n' to depict orbital size. Schrödinger's three-dimensional model required three quantum numbers—principal (n), angular (l), and magnetic (m)—to characterize the size, shape, and orientation of electron orbitals.
The principal quantum number (n) defines the orbital size, indirectly reflecting energy. Angular quantum number (l) signifies orbital shape, distinguishing between spherical (l=0), polar (l=1), and cloverleaf (l=2) shapes. The magnetic quantum number (m) is introduced to pinpoint orbital orientation. Rules govern the combinations of these quantum numbers: n, l, and m are integers with specific constraints.
Shells, composed of orbitals sharing the same principal quantum number, are further divided into subshells with the same angular quantum number. Subshells, denoted by characters like s, p, d, and f, elucidate the electron configuration. Each shell houses orbitals whose number is the square of the principal quantum number, and each subshell contains 2(l)+1 orbitals.
Exceptions arise in chromium and copper, where one electron moves from 4s to 3d, defying the predicted electron configuration. However, the Aufbau principle generally guides the orderly filling of orbitals, defining elements' electron configurations and aligning with the periodic table's structure. The relative energies of orbitals, governed by size and shape, determine electron arrangement, showcasing the remarkable predictive power of quantum principles in understanding atomic behavior.
Quantum tunneling is a phenomenon that allows particles to pass through potential barriers that would be impossible with classical physics. In classical mechanics, a particle requires enough energy to overcome a barrier. Quantum mechanics, on the other hand, introduces the concept of wave-particle duality, where particles also exhibit wave-like properties. These wavefunctions go beyond the physical barrier, creating a probability that the particle can "tunnel" through it, even when it lacks the energy to do so conventionally.
This occurs because of the probabilistic nature of quantum mechanics. If a particle’s wavefunction has a nonzero amplitude on the other side of the barrier, there is a chance the particle will appear there. While this seems counterintuitive, tunneling has been experimentally validated and is essential to many natural and technological processes.
Quantum tunneling is vital in the functioning of electronic devices, particularly in tunnel diodes and transistors. In these devices, electrons tunnel through thin insulating layers, allowing for current flow at nanoscale dimensions.
Quantum tunneling plays a major role in the rates of certain chemical reactions—especially those involving small particles like protons or electrons. In classical terms, a reaction might seem impossible at low temperatures due to insufficient thermal energy to overcome activation barriers. With quantum tunneling, on the other hand, particles can bypass these barriers, allowing reactions to proceed even when conditions aren’t ideal.
In molecular systems, for example, tunneling impacts hydrogen transfer—crucial for processes like isomerization and bond formation. By allowing particles to "tunnel" through energy barriers, reactions occur faster than predicted by classical models.
Quantum entanglement is where multiple particles become linked in such a way that the state of one particle is immediately connected to the state of another—no matter how far they are from each other. This means that measuring the state of one particle determines the state of the other. In comparison, classical ideas of locality state that objects can only influence one another through direct interaction or signals traveling no faster than the speed of light.
The concept of entanglement was introduced by physicists like Albert Einstein, Boris Podolsky, and Nathan Rosen in their famous EPR paradox. This was later mathematically formalized by John Bell. Experiments validating entanglement—such as those by Alain Aspect in the 1980s—confirmed that quantum mechanics allows for non-local correlations, challenging the classical view of a deterministic universe.
In quantum information science, quantum entanglement opens avenues for technologies that use the properties of quantum systems. Entangled qubits let quantum computers perform computations in parallel, boosting their processing power for certain tasks compared to classical computers.
Quantum computing: As a cornerstone of quantum computing, entanglement allows qubits to function in correlated states. This improves computational capabilities for solving complex problems far beyond the reach of classical computers.
In quantum mechanics, significant advancements made by quantum computing companies are reshaping our understanding of chemistry. Quantum chemistry, a blend of quantum mechanics and chemistry, plays a crucial role by providing a detailed look into how molecules bond. This involves figuring out which combinations of molecules are more likely to happen and the specific amounts of energy involved in these processes.
Quantum mechanics, which started with a focus on orbitals, has evolved into a versatile tool beyond just studying electron behavior. It's become quite practical, especially in predicting and explaining chemical reactions. Quantum mechanics helps chemists anticipate how different chemicals will interact by clearly showing which molecular setups are energetically favored.
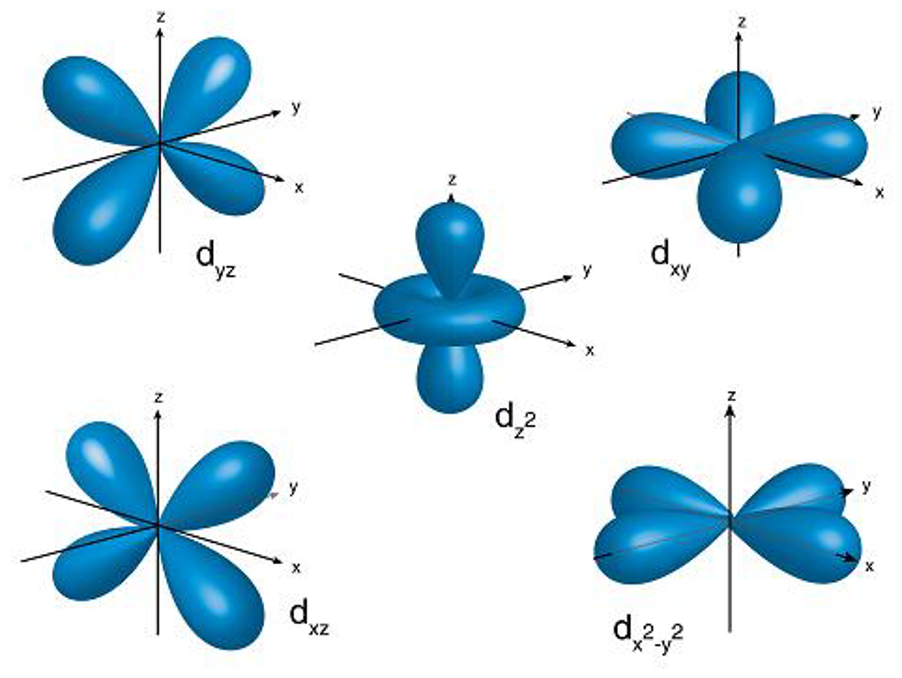
So, this quantum mechanical model isn't just a theoretical concept – it's a valuable tool for chemists. It guides us from understanding the basics to predicting and controlling real-world chemical reactions. As we advance our knowledge and technology, the partnership between quantum mechanics and chemistry keeps opening up new possibilities in materials science, catalysis, and the development of new medicines.

For those keen on delving into the quantum realm, BlueQubit offers a unique opportunity. Scientists and quantum enthusiasts alike can utilize the quantum computing software platform to experiment and test complex problems in quantum computing. BlueQubit provides a space where individuals can practically engage with the principles discussed here, making quantum experimentation more accessible. BlueQubit can help us navigate the interesting link between chemistry and quantum physics.
When studying the behavior of tiny particles like electrons, quantum mechanics is similar to taking a deep dive into the microscopic world. Quantum mechanics provides a degree of uncertainty in contrast to traditional physics, which deals with exact measurements. Imagine electrons not following fixed paths but existing in areas called orbitals with a 90% chance of being there. The Schrödinger equations' description of this probability-based strategy aids in our comprehension of electron behavior.
Now that we have observed the development of quantum understanding from Bohr to Schrödinger, we see how this knowledge isn't just theoretical.
And here's where BlueQubit steps in. It's a quantum simulation platform where scientists and quantum enthusiasts can practically engage with these principles. If you're curious about the fusion of chemistry and quantum physics, BlueQubit provides hands-on experience.
The quantum mechanical model was developed through the collaboration of several physicists in the early 20th century. In 1900, Max Planck initiated the concept by introducing the idea of quantized energy levels. Niels Bohr then expanded on this discovery with his model of the hydrogen atom in 1913. Later, scientists like Werner Heisenberg, Erwin Schrödinger, and Paul Dirac solidified the model by formulating the mathematical foundations of quantum mechanics.
The quantum mechanical model predicts the behavior and properties of particles at atomic and subatomic scales. It offers a probabilistic framework for scientists to understand electron positions and energies. The model accounts for the wave-particle duality of matter, allowing electrons to exist in superpositions of states. It also predicts quantum phenomena such as tunneling, entanglement, and the uncertainty principle, which classical physics cannot explain.
Quantum mechanics is a fundamental theory in physics that describes the physical properties of nature at the scale of atoms and subatomic particles. It introduces concepts like wave-particle duality, quantization of energy, and the uncertainty principle. The theory makes it possible to predict and understand phenomena such as electron orbitals, atomic emission spectra, and the behavior of particles in potential wells. Quantum mechanics can be applied in fields like chemistry, material science, electronics, and quantum computing.
The term "four quantum models" typically refers to the four quantum numbers used to describe the quantum state of an electron in an atom. These are the principal quantum number (n), the azimuthal (angular momentum) quantum number (l), the magnetic quantum number (mₗ), and the spin quantum number (mₛ). Together, these quantum numbers describe an electron's position and behavior within an atom.